Key Partners
Established in 2016 in partnership between Duchenne UK and the John Walton Muscular Dystrophy Research Centre at Newcastle University, the DMD Hub is the UK clinical research network for Duchenne muscular dystrophy. The DMD Hub exists to enable and accelerate delivery of clinical trials in the UK for the benefit of patients, clinical sites and industry.
Duchenne UK

Duchenne UK was founded by two parents, Alex Johnson and Emily Reuben, after their sons were diagnosed with Duchenne muscular dystrophy. They joined together to form Duchenne UK in 2016.
As the leading Duchenne muscular dystrophy (DMD) charity in the UK, Duchenne UK aim to end Duchenne's devastating impact. They connect scientists, the pharmaceutical industry, the NHS and families to advance and accelerate medical research, while bringing everyone affected by DMD the care and support they need.

John Walton Muscular Dystrophy Research Centre
Launched in 2014, the John Walton Muscular Dystrophy Research Centre brings together and consolidates Newcastle’s world-leading research and care for neuromuscular diseases. The team is based at the Institute of Genetic Medicine, Newcastle University. The centre is structured around five key activities: clinical care, clinical research, diagnostics, basic research and strategic partnerships and networking.
STEERING COMMITTEE

Emma Heslop
Emma Heslop is the DMD Hub manager. Emma is based at the John Walton Muscular Dystrophy Research Centre, in a position funded by Duchenne UK. Emma trained as a biological anthropologist at Durham University and was awarded an MSc (Research) in 2005. She has been part of the John Walton Muscular Dystrophy Research Centre at Newcastle University since October 2006, when she joined the TREAT-NMD Neuromuscular network of excellence, as an assistant project manager.
Emily Reuben
Emily is the co-founder and joint CEO of Duchenne UK. Emily set up Duchenne Children’s Trust in 2012 after her son was diagnosed with Duchenne muscular dystrophy. Before that she was a reporter for Channel 4 News and CNN international. Emily has advocated on behalf of patients around the world. She’s spoken many times in the Houses of Parliament. Emily is a member of the ABPI’s eight-person Patient Advisory Council, and sits on the Steering Group for the ABPI’s Patient Organisation Forum. She is also a member of the MHRA’s Patient Group Consultative Forum, as well as the Steering Group for the regulator’s new Innovative Licensing and Access Pathway (ILAP).
Alex Johnson
Alex is the co-founder and joint CEO of Duchenne UK. Alex founded the charity Joining Jack with her husband,ex-rugby league player Andy Johnson, following their son Jack’s diagnosis with DMD in 2011. Alex is a board member of the World Duchenne Organisation (WDO) and has helped organise international awareness events like the World Duchenne Awareness Day. Alex has represented the Duchenne community by speaking at international conferences around the world, workshops and meetings with the FDA, EMA, MHRA, NICE and at the Houses of Parliament. Alex was honoured to join the steering committee that helped organise a workshop with the EMA on exon skipping. Following the meeting the steering committee published an article in the Lancet. Alex completed training at a Eurordis summer school to become a trained patient advocate and is also a member of the MHRA patient group consultative forum.
Professor Vol ker Straub
ker Straub
Professor Straub is the Deputy Dean, Harold Macmillan Professor of Medicine and Professor of Neuromuscular Genetics at the Institute of Translational and Clinical Research at Newcastle University. One of Professor Straub’s main interests in muscle diseases is around translational research. He was the co-founder of the EU FP6 funded network of excellence for genetic neuromuscular diseases, TREAT-NMD, which he coordinated together with Kate Bushby.
Professor Michela Guglieri
Professor Guglieri is a Senior Clinical Lecturer and Honorary Consultant at Newcastle University. She leads the clinical research team which is currently involved in over 30 studies over the past 5 years. Duchenne UK collaborated with 5 patient organisations to award a 5-year lectureship to Professor Guglieri in 2015.
 Dr Giovanni Baranello
Dr Giovanni Baranello
Dr Baranello has been involved as principal investigator in numerous neuromuscular clinical trials, predominantly in the areas of Spinal Muscular Atrophy and Duchenne Muscular Dystrophy. He has also gained significant independent grant support and funding to undertake research in neuromuscular diseases. His main area of research relates to translational research in neuromuscular conditions, development of outcome measures, natural history studies and long-term studies in patients receiving novel treatments.
 Dr Anne-Marie Childs
Dr Anne-Marie Childs
Dr Childs has been working as a Consultant Paediatric Neurologist in Leeds since 2001 when she became the Lead for Children’s Neuromuscular Services. She is committed to delivering high-quality care to all patients with neuromuscular disorders in Yorkshire. Her clinical research interests include MR imaging and rare disease phenotyping, although her predominant research involvement is now in relation to DMD and SMA.
Jon Hastie
Jon Hastie lives independently in Reading supported by a team of Personal Assistants. He has a background in policy, communications and engagement and has worked in local government, the charity sector and the pharmaceutical industry. Jon founded DMD Pathfinders in 2014 with late friends Mark and Robert, and was CEO of Pathfinders from 2016-22, managing their transition to Pathfinders Neuromuscular Alliance. Jon has Duchenne Muscular Dystrophy. His hobbies are gaming, nature walks and movies, and he enjoys writing blogs.
DMD Hub Sites
Centres of Excellence:
DMD Hub Sites:
Working in Partnership
The DMD Hub is working closely with the North Star Network, TREAT-NMD, NIHR, and Duchenne Muscular Dystrophy Charities.
Funding
Seed funding for the DMD Hub comes from Duchenne UK. Other funding has come from Alex's Wish, Jack's Mission and some other DMD charities. Duchenne UK also fund some posts in collaboration with the NHS.
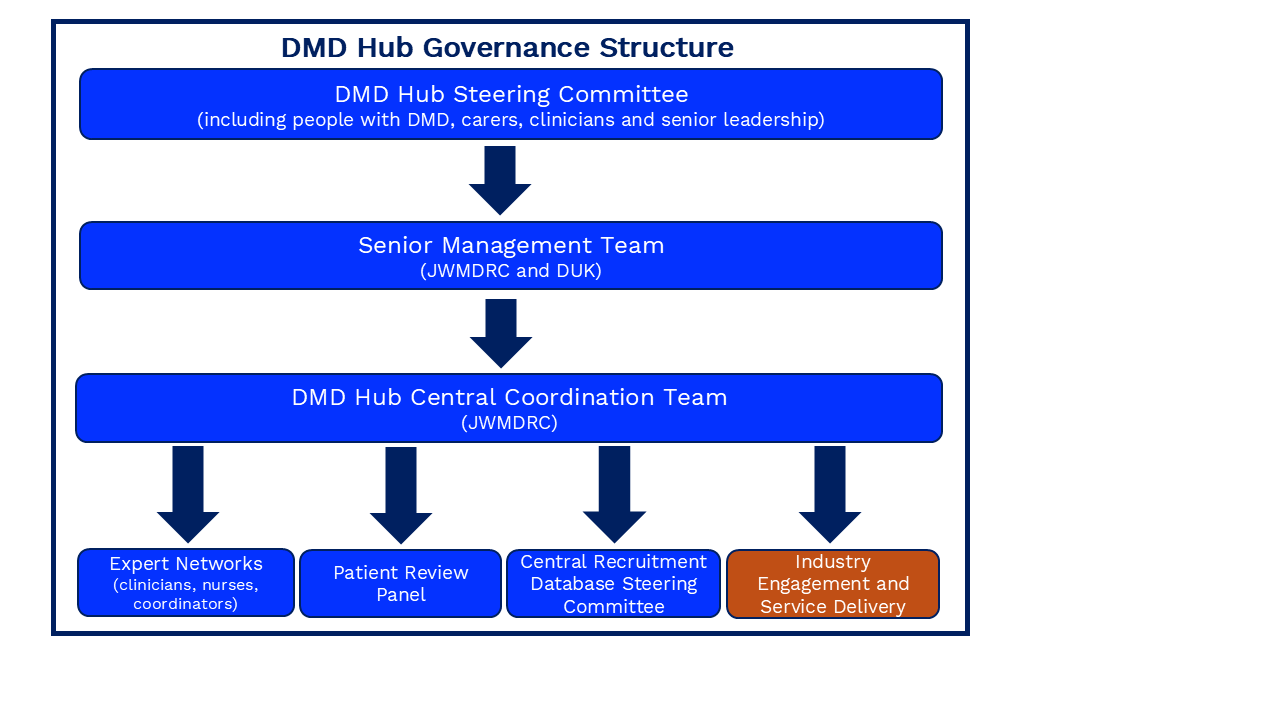
DMD Hub Governance Structure
Our Staff Network

I joined the DMD Hub as a Clinical Trial Coordinator in May 2021. I have worked at The Robert Jones and Agnes Hunt Orthopaedic Hospital since September 2020 where I originally joined the research department as a placement student for my university degree. I am expected to graduate from my Biomedical Science degree in May 2022, where I would have completed 4 years of study. I work on neuromuscular studies within the department and coordinate the DMD studies we are currently running.

My role is to make sure that all trial visits are carried out within the trial timetable and that parents are given appointment information for their visits. I liaise with the clinical research team to organise the appointment dates and make sure that any tests that are required for the trial visit are arranged for the same day, as well as arranging travel for patients and their families. I am also responsible for data input onto the various clinical trial databases, answering any queries from the trial sponsors and accurate upkeep of the trial Master Site Files for each clinical trial taking place in Glasgow. In addition, I organise monitoring and site inspection visits.

Ellie Drummond - Clinical Trial Coordinator
Newcastle University and The Newcastle upon Tyne Hospitals NHS Foundation Trust
As a Clinical Trial Coordinator at the JWMDRC, my role is to support the set-up and coordination of our clinical research portfolio, which includes clinical trials of an investigational medicinal product and gene replacement therapy.
I support the studies at a very early stage from feasibility to costing negotiations to the set-up and monitoring delivery of research, ensuring research studies that are conducted adhere to Good Clinical Practice (GCP).
I work closely with teams within Newcastle University, the Clinical Research Facility and the Newcastle-upon-Tyne Hospitals NHS Foundation Trust, together with Sponsors and Contract Research Organisations across the world to get trials up and running as efficiently as possible in order to facilitate the delivery of world class research.

Bethany Bashford-Chuchla - Paediatric Research Nurse
Great Ormond Street Hospital NHS Foundation Trust
I qualified as a children’s nurse in 2016 and have worked in a range of settings including a general paediatric and surgical ward, a busy trauma A&E and abroad. I recently joined the clinical research facility at GOSH and now work on a number of studies within the renal and neuromuscular portfolio.

I have previously worked at GOSH as a data manager on a number of ongoing clinical trials. I have recently joined the GOSH neuromuscular research team as a coordinator and will be working on the set up and delivery of a range of studies within the portfolio.


I’m a Senior Research Coordinator at Great Ormond Street managing a portfolio of neuromuscular studies. This involves setting up, day to day management of and closing of clinical trials.

Marta is an enthusiastic and dedicated individual with extensive and in depth experience in the coordination and management of paediatric clinical trials, from first in human/child and early phase studies to gene therapy trials.

After completing a degree in Children’s nursing in 2001, Donna started her career with the Leeds Teaching Hospital in children’s medicine. In 2007 she achieved a Community Specialist Practitioner First class Degree with Honours, but instead of moving fully into a community role, she chose to become a Children’s Nutrition Nurse Specialist. This enabled her to combine her experience, skills and passion for working in an acute setting with the autonomy and the community aspects of supporting children and families to manage complex nutritional health needs at home.
Donna has been a Children’s research nurse at Leeds Children’s Hospital since May 2017 and has worked across many areas of paediatric research, utilising her background in general medicine. In the last few years she has focused more on Neuromuscular clinical trials and enjoys working alongside Dr Childs and the Neuromuscular team at Leeds to deliver high quality research and patient care.

Laura Thrasyvoulou - Paediatric Research Nurse
University Hospitals Birmingham NHS Foundation Trust (Birmingham Heartlands Hospital)
As the Paediatric Research Nurse for DMD and SMA, based at Birmingham Heartlands Hospital, part of the University Hospitals Birmingham NHS Foundation Trust, I am involved in the set-up, running and close down of clinical trials both commercial and academic for the DMD and SMA population. There are 3 Paediatric Research Nurses and a dedicated Data Manager that been providing high quality research for over 6 years. Our commercial trial profile has increased dramatically over the last 4 years and continues to do so. We pride ourselves on providing a welcoming and safe environment for the patients and their parents / caregivers. We are constantly striving to improve our research environment to ensure that the patients are comfortable and happy, with the aim of having a positive experience whilst enrolled on a research study.

Katie Price - Paediatric Research Nurse
University Hospitals Birmingham NHS Foundation Trust (Birmingham Heartlands Hospital)
I graduated as a Paediatric Nurse in 2012 and worked in children’s Intensive care for over 7 years. Here I gained a particular interest in research and went onto work in their research team for 2 years before I started working for Birmingham Heartlands Hospital, part of University Hospitals Birmingham NHS Foundation Trust as a Paediatric Research Nurse in 2019. I work on a variety of trials that cover several specialities but the majority of our studies are academic and commercial trials in the DMD and SMA population. We strive to provide high quality research within our team.


James completed his paediatric nurse training in 2005 in Manchester and spent 3 years working at the burn's unit at Manchester Children's Hospital. He relocated to Shrewsbury in 2008 and spent time working on neonates before working for 6 years full time at Hope House Children's Hospice providing specialist palliative care to children and young adults. In 2015 James took on a new research post at Shrewsbury and Telford Hospital Trust and spent 5 years developing the paediatric portfolio as well as supporting many other specialities within research. He continued and still continues to work one day a week at hope house clinically and is passionate about supporting families clinically and driving research.
Fay has 19 years of experience as a Neonatal Acute transport team nurse and has brought a wealth of experience from this previous post.
She joined the Neuromuscular team in November 2017 and is the first point of contact for family queries.
She liaises with all the families, organises the joint clinics, study days and internal meetings within the department. She also helps the physiotherapists with their assessments in the neuromuscular clinics.

Jen has worked as a Neuromuscular Nurse Specialist within the team for the past six years. Her current remit is to organise and undertake clinical assessment at North Star clinics, co-ordinate patient care (both acute and out-patients) and is the trial coordinator for our current FOR-DMD trial. Jen is a nurse prescriber and is currently studying for an MsC in Advanced Nursing Practice. She is in the process of completing an audit, investigating patient education and understanding of IM Hydrocortisone in steroid-treated DMD boys.



Jayne Banks - Senior Research Nurse
Newcastle University and The Newcastle upon Tyne Hospitals NHS Foundation Trust
I am a paediatric research nurse and I have worked on the CRF since 2012 delivering early phase clinical trials to both children and adults. The disease areas I have worked with on clinical trials have included DMD, SMA, Achondroplasia, Haemophilia and JIA. Other clinical trials in set up are looking at epilepsy, mitochondrial disease and autism treatments. We enrol participants regionally, nationally and internationally.
We have a dedicated paediatric area within the department and aim to deliver family centred, holistic care working with both academic and pharmaceutical partners to develop new treatments for chronic and rare diseases. We have delivered the first UK Pfizer DMD gene therapy dose in May 2021 and I am currently involved in the set-up of a number of other gene therapy studies.

Rebecca graduated from Edge Hill University in 2013, and was awarded with a bachelor of Science degree in Children’s Nursing. She received her first nursing post at a Alder Hey Children’s Hospital, working as a staff nurse on a medical specialty ward from 2013-2018. She then joined the clinical research team in 2018 and was assigned to the DMD research post in March 2020, before she was seconded to assist with COVID-19 research studies.
Rebecca is currently working with the Duchenne Muscular Dystrophy Research team. She co-leads on numerous DMD trials and works alongside a team of DMD research professionals. Her role helps to assist the study sponsors in finding better and safer treatments for children and young people with DMD, and hopefully in the future to find a cure.

Tim has been appointed as a Duchenne muscular dystrophy Research Nurse at Alder Hey. He qualified as a staff nurse at Alder Hey in 2003 and worked on the regional oncology unit at Alder Hey.
He became a research nurse at the Clinical Research Facility in 2012, where he has lead on multiple early phase research studies, including oncology studies, studies in theatre, and gastroenterology studies amongst others. Also, he has led on 2 studies that recruited the first global patients to the study.
Away from work he is a family man and spends most of his weekends watching his children play multiple sports! He is a keen cyclist and plays football socially once a week.

Paula Sugden - Paediatric Research Nurse
Lancashire Teaching Hospitals NHS Foundation Trust; Royal Preston Hospital
Paula works as a research nurse in the Child Health Department at Royal Preston Hospital. She has a wide role in the set up and management of research studies in children, and advocating research in our trust. She has been closely involved in the development and running of a trial looking at a new device to help children who wet the bed, and is about to start an exciting study looking at determinants of activity in children with muscle conditions.
Susanne McKenzie - Specialist Neuromuscular Physiotherapist
Royal Hospital for Children, Glasgow
Susanne is a Specialist Neuromuscular Physiotherapist, working with the Neuromuscular team at the Royal Hospital for Children, Glasgow, since 2014. As part of her role, Susanne is involved with clinical trials, as a clinical evaluator. In 2020, she is working on the VISION-DMD and TAMDMD trials, and is also the research assistant for FOR-DMD and SCOT-DMD. The team are preparing to be involved in the SAREPTA-ESSENCE trial later in 2020. In addition to trials, Susanne assesses and treats neuromuscular patients along with the multi-disciplinary team. This includes out-patients, in-patients and community visits.

Rebecca has been working with the Neuromuscular Physiotherapy team in Glasgow since March 2020. Her post was initially funded by the DMD Hub to support the team’s trial capacity and has now been made substantive in the NHS. She has extensive experience in a range of specialist rotations in both adults and paediatrics, in hospitals in and around Glasgow. She has worked in the Royal Hospital for Children in Glasgow since 2018. Rebecca has worked as a research assistant on the TAMDMD, VISION-DMD and Sarepta-ESSENSE drugs trials and clinical evaluator in the ULYSSES trial. Rebecca also supports the DMD clinics and works with the wider neuromuscular population to support the team in pursuing further upcoming trials.

Sarah has worked at the Royal Hospital for Children Glasgow for fifteen years, and as part of the Neuromuscular Physiotherapy Team for twelve of these years. This post involves working alongside the neuromuscular multidisciplinary team, assessing and managing patients at the out-patient clinics and as in-patients on the wards. Supporting patients and their families in their homes and schools is also a key part of the post. Clinical research trials have become an increasing aspect of the role and she is a Clinical Evaluator for several trials. She completed an MSc in Advanced Practice in 2024.

Marina Di Marco - Consultant Neuromusuclar Physiotherapist
Royal Hospital for Children, Glasgow
Marina Di Marco is a Consultant Neuromuscular Physiotherapist based in the Children's Hospital, Glasgow. She has worked in the field of neuromuscular disorders for over thirty years and works with children and their families living with muscle wasting conditions across the West of Scotland. Marina has a clinical, research and teaching remit, and leads the neuromuscular physiotherapy service in Glasgow and the West of Scotland.
Marina participates in training physiotherapists for research trials globally and presents on standards of care and physiotherapeutic interventions at international workshops.

Lindsey has over 20 years experience as a member of the Leeds Neuromuscular team. She works as part of the MDT but also runs Physio led clinics seeing patients for follow-up. For the last 10 years, she has also been working as the clinical evaluator in an increasing number of research studies that have opened at our centre completing a wide range of motor and respiratory assessments on young people.
Lindsey is an active member of the Association of Paediatric Chartered Physiotherapists and has chaired the Neuromuscular Specialist Group. She is a member of the Northstar and SMA Reach physio networks.
She is an ATOM trainer for physiotherapists participating in clinical trials.

Hayley Ramjatten - Specialist Neuromuscular Physiotherapist
Oxford University Hospital and University of Oxford
Hayley is a Specialist Neuromuscular Physiotherapist, at Oxford University Hospitals. She sees a large cohort of patients with neuromuscular conditions, who access Oxford as their tertiary centre for neuromuscular and respiratory care.
She supports the Oxford diagnosis specific multidisciplinary clinics, which run on a weekly basis and also provides outreach support to general neurology clinics and offers community and hospital visits as required.
Within her role she supports patients in their orthopaedic and respiratory management, offering input during planned and unplanned admissions and linking in with other members of the wider multidisciplinary team and community therapists.
She is also a part of the Oxford Congenital Myasthenia Service. This is a nationally funded service, which includes both paediatric and adult patients. She is currently working on projects in collaboration with other specialist physiotherapy colleagues, on the application of outcome measures and exercise advice in this patient cohort.

Charlotte Lilien - DMD-Hub Advanced Physiotherapist Neuromuscular Service
Oxford University Hospital and University of Oxford
Since my Masters degree in Physiotherapy obtained in June 2013, I started to work in September 2013 as Physiotherapist at the Institute of Myology; a centre specialized in care and research in adult and paediatric neuromuscular diseases. Over the course of my career, I have participated in the creation of the I-Motion Institute, and was the Physiotherapy Coordinator for an international natural history study in Myotubular Myopathy.
I am also a part of the ActiMyo team, composed of engineers and clinicians. This innovative device uses magneto-inertial sensors to permit movement, gait and activity analysis in uncontrolled environments, and one of its application has been qualified by the European Medical Agency as a clinical endpoint in clinical trials in Duchenne muscular dystrophy. My role is to give a physiotherapy point of view and input, and to train the physiotherapists conducting the clinical trials. Since 2018, I’m a Master trainer and consultant for scales and outcome measures used in neuromuscular clinical trials and clinics.
After more than six years of collaboration with Laurent Servais in Paris, I decided to join his team within the MDUK Oxford Neuromuscular Centre, a partnership between the University of Oxford and Muscular Dystrophy UK to drive the development of new therapies and increase national clinical trial capacity in neuromuscular diseases. .

Nicholas Emery - Neuromuscular Clinical Specialist Physiotherapist
Robert Jones and Agnes Hunt Hospital
I qualified from Keele University in 2005 with a first in Physiotherapy. I started working at the Neuromuscular Centre in Winsford, treating and managing adults with NMD. This involved a range of interventions from stretching programmes, musculoskeletal assessments and treatment through to hydrotherapy and exercise prescription.
In 2009, I started work at the RJAH Orthopaedic Hospital working alongside Dr Quinlivin as part of the NMD MDT. This again involved management of adults with NMD but now also paediatric patients. In addition, my role expanded to include being involved in NMD research trials. I also worked in two further departments:
1. Orthotic Research and Locomotion Assessment Unit (ORLAU), a specialist department assessing abnormal gait and provision of various interventions to improve functional walking.
2. Alice Ward, which involved the treatment and rehabilitation of paediatric patients with a range of conditions - CP, NMD, chronic pain, CFS etc.
I have also undertaken further study at Keele University obtaining a Postgraduate Diploma (Physio) in 2012. I have attended a variety of external conferences concerned with the care and management of NMD patients.

Sinead Warner - Neuromuscular Physiotherapist
Royal Manchester Children's Hospital, Manchester University Hospitals NHS Foundation Trust
Sinead Warner completed her physiotherapy degree in Manchester and following this moved to New Zealand, where she completed rotational posts including paediatric specialities. On returning to Manchester, Sinead worked as a paediatric community physiotherapist where she developed an interest in neuromuscular conditions. Sinead now works as a highly specialised physiotherapist within the Neuromuscular team.

Jennie Sheehan - Clinical Specialist Paediatric Physiotherapist
Evelina London Children's Hospital
Jennie is a chartered and state-registered physiotherapist with many years of experience in paediatrics. She has worked for most of her career in paediatric neuromuscular disorders, working at a number of specialist centres, including The Hammersmith Hospital with Professor Dubowitz, then Professor Muntoni. When working at Guys Hospital, she was involved in both the Paediatric and Adult Neuromuscular clinics. For the last 11 years, she has been at The Evelina London Children's Hospital. She also has experience as a Paediatric Community Physiotherapist, and is therefore very aware of the challenges that are met by therapists working in different environments.
She works as part of the MDT team at The Evelina London, carrying out standardised assessments, providing clinical management and advice to families and local physiotherapists. She also works closely with the orthopaedic and respiratory physiotherapy teams, and the orthotists.
Jennie was one of the developers of the NorthStar Ambulatory Assessment for DMD, which after publication, became widely recognised and adopted as the standardised assessment of choice for many research studies in DMD, both nationally and internationally, and is also an essential part of the North Star national database. She is the physiotherapy representative on the National North Star Steering Committee.
Jennie has been involved in a number of research projects and publications - the most recent being the national study on hydrotherapy in DMD, as the independent assessor of prescription suitability and compliance. She has set-up, and continued to run, the South East Physiotherapy Network for NMD. She is a member of APCP and the APCP Neuromuscular group, and was previously a Committee member.
Jennie is the lead for a small team of specialist physiotherapists in Neuromuscular Disorders at Evelina London.
Cath qualified from Coventry University in 2004 with a BSc Hons Physiotherapy. Cath completed her rotations at Peterborough District Hospital. In 2006, Cath began working as a Community Paediatric Physiotherapist in Waltham Forest, before moving to her current post as a Community Paediatric Physiotherapist at Cambridgeshire Community Services NHS Trust in 2009.
Cath developed a special interest in Neuromuscular Disorders from her time working with this client group during her community paediatrics placements. Cath began working in the Addenbrookes Neuromuscular Service from when it was first formed, developing the physiotherapy role and provision within the service over the years. Cath has provided support and training to community therapists within the region, regarding the management of neuromuscular disorders and she has been involved in the physiotherapy role for the SUMMIT clinical trial.
When not in the clinic, Cath works for Cambridgeshire Community Services (CCS NHS Trust) as a Senior Community Paediatric Physiotherapist. This role is primarily a clinical role supporting CYP with complex needs in the community, including those with neuromuscular conditions.


Rosanna Rabb - Paediatric Neuromuscular Physiotherapist
University Hospitals Birmingham NHS Foundation Trust (Birmingham Heartlands Hospital)
I graduated in 2001 and completed my junior rotations at Hammersmith and Charing Cross Hospital, London. Here, I gained experience working with one of the leading neuromuscular services and it started my special interest in this field. I then went on to specialise as a senior paediatric physiotherapist at Hammersmith hospital in 2003, which also involved working within the paediatric physiotherapy department at St Mary's Hospital in London, and gaining experience within both the community and acute paediatric physiotherapy.
After traveling the world for a year, I then came back to England to work at Chelsea and Westminster hospital as a Senior Paediatric Physiotherapist before relocating to Birmingham. Initially I worked for a year as a community physiotherapist, both within the special needs schools and the child development centres. I then moved to Birmingham Children’s Hospital where I worked for 9 years and specialised in the field of neurology, with a particular interest in neuromuscular. In 2017, I was appointed to my current role at Birmingham Heartlands Hospital, as a Paediatric Neuromuscular Physiotherapist.
Heather McMurchie - Paediatric Neuromuscular Physiotherapist
University Hospitals Birmingham NHS Foundation Trust (Birmingham Heartlands Hospital)
I work at Birmingham Heartlands Hospital as a Specialist Neuromuscular Physiotherapist in the Regional Paediatric Neuromuscular Clinic. I undertake assessments and support the local physiotherapy teams with the treatment of our patients. I am involved in a number of national and international research studies for the treatment of DMD and SMA.
I qualified as a Chartered Physiotherapist in 1992 and commenced work at Birmingham Heartlands Hospital as a junior physiotherapist undertaking a variety of rotations. I have worked in paediatrics since 1996 and started specialising in paediatric neuromuscular conditions in 1998. During this time the management of neuromuscular conditions has improved significantly, I have enjoyed being part of this development, and I am looking forward to the advances.

Dionne Moat - Neuromuscular Research Physiotherapist
Newcastle University and The Newcastle upon Tyne Hospitals NHS Foundation Trust
I am a physiotherapist and have been qualified for 15 years. My background is neuro-rehabilitation and I joined the Newcastle Muscle team is 2016. I work with both children and adults with neuromuscular disease in clinical trials. I am currently studying for a Masters in Clinical Research at Newcastle University.

Jassi Amritpal Singh Sodhi - Neuromuscular Research Physiotherapist
Newcastle University and The Newcastle upon Tyne Hospitals NHS Foundation Trust
I am a neuromuscular research physiotherapist based at the John Walton Muscular Dystrophy Centre. I joined the team in March 2017. Prior to my current role I have worked as a senior physiotherapist working with people with learning and developmental disabilities.
My current role allows me to work across both clinical and research strands. I am currently involved with multiple clinical trials in Duchenne muscular dystrophy, as well as a natural history study in dyferlinopathy.

Robert Muni Lofra - Consultant Neuromuscular Physiotherapist
Newcastle University and The Newcastle upon Tyne Hospitals NHS Foundation Trust
I am a Neuromuscular Physiotherapist at the John Walton Muscular Dystrophy Research Centre in Newcastle upon Tyne (UK). I started working with neuromuscular patients back in Barcelona in Hospital Sant Joan de Déu as a Paediatric Physiotherapist. In 2014, I moved to the JWMDRC to work exclusively in NMD. My professional activity covers provision of clinical management and research activity in Neuromuscular diseases with a special interest in SMA. Robert is currently undertaking a PhD.

Meredith James - Clinical Specialist Neuromuscular Physiotherapist
Newcastle University and The Newcastle upon Tyne Hospitals NHS Foundation Trust
Meredith James is a Clinical Specialist Neuromuscular Physiotherapist at the John Walton Centre for Muscular Dystrophy Research at Newcastle, where she is involved in clinical and research activity for children and adults with Neuromuscular disorders.
Meredith moved to the UK in 2004 from Australia and worked in a variety of locations before joining the Oswestry Muscle team in 2006 and coming to Newcastle in 2012.
Clinically, Meredith is responsible for the physiotherapy management of both children and adults with neuromuscular diseases. In particular, her interests are in assessment, orthotics, outcome measures and research into these areas.
In her clinical research capacity, Meredith is responsible for the clinical evaluation of children and adults involved in natural history and clinical trials, as well as the development of clinically meaningful, reliable and sensitive outcome measures for NMD.

Sarah Gregson - Specialist Neuromuscular Physiotherapist & Research Physiotherapist
Alder Hey Children's NHS Foundation Trust
Originally from Cork, Ireland, Sarah moved to Bristol in September 2000 to study physiotherapy at the University of West of England in Bristol. After qualifying in 2003, Sarah took up her first post as a junior physiotherapist in Hope Hospital in Salford until moving to Alder Hey Children’s Hospital, Liverpool in 2006 as a Senior Physiotherapist. She worked across all acute and community paediatric specialities gaining an interest in Neurology. In 2010, Sarah started a MRes degree at the University of Liverpool, which allowed her to complete research projects of interest to her and her area of clinical work. After a break for maternity leave, Sarah moved into the Neuromuscular Physiotherapy team in 2012, initially as a temporary position and then became a permanent member of the team in 2014. In 2015, she completed her MRes in clinical Science with distinction. Having gained a keen interest in research during her MRes studies, Sarah took the opportunity to take up the research physiotherapist post as part of the Alder Hey Neuromuscular team, in collaboration with Duchenne UK.

Candiss Argent - Paediatric Research Physiotherapist
Lancashire Teaching Hospitals NHS Foundation Trust; Royal Preston Hospital
Candiss is a Paediatric Research Physiotherapist with a background in community paediatric physiotherapy. She has recently joined the research team at Lancashire Teaching Hospitals to help set up and run Dr DeGoede’s DMD Hydro study. Prior to that Candiss developed her specialist skills working for a children’s charity that provided assessments and treatments for children with complex needs, gaining post graduate qualifications in Bobath and Sensory Integration. She then accepted an NHS post as a lead physiotherapist in a special school alongside holding a varied caseload in paediatrics, including respiratory. She completed a secondment in Grand Cayman as a Locum Paediatric Physiotherapist before returning to her previous NHS post. Candiss has always been keen to be involved in research and so found this research post at Royal Preston Hospital very exciting. This interest started at University where she was a participant in an educational study and has continued since qualifying as she regularly consents to participate in studies that require physiotherapists to participate.
